Inner Sphere Electron Transfer - The changes that take place at each center when the electron count. The inner sphere mechanism shown diagrammatically above involves the joining of two complexes via a bridging ligand and the loss of another ligand to accommodate this steric change.

Molecular Orbital Theory And Bond Order Molecular Electron Configuration Theories
Electron transfers that occur via ligands shared by the two metals undergoing oxidation and reduction are termed inner sphere electron transfers.

Inner sphere electron transfer. The inner sphere mechanism is important because atom transfer bond breakingformation can be concomitant with electron transfer. Here a ligand bridges the oxidant and reductant reactants during the. 1 The bridge connects the two metals.
This complex can then transfer electron density via the bridging ligand from one metal centre to another. Processes of simple chemistry. Roughly 450 550 light-years How many mechs are in a lance.
Inner sphere or bonded electron transfer is a redox chemical reaction that proceeds via a covalent linkagea strong electronic interactionbetween the oxidant and the reductant reactants. Inner sphere electron transfer. Inner sphere electron transfer and Activation energy See more Antimony tetroxide.
How big is the inner sphere. In coordination chemistry a bridging ligand is a ligand that connects two or more atoms usually. En la transferencia de electrones de la esfera.
In the outer sphere model the donor or acceptor and the tightly bound solvation shells or the complex ligands were considered to form rigid structures which do not change in the course of electron transfer. If playback doesnt begin shortly try restarting your device. Inner sphere electron transfer in English-Spanish dictionary.
Antimony tetroxide is an inorganic compound with the formula Sb2O4. Inner sphere mechanism of electron transfer is a redox chemical reaction that occurs via a covalent linkage between oxidant and reductant reactant of the reaction. Four How many mechs are in BattleTech.
Search over 14 million words and phrases in more than 490 language pairs. Both are important for photosynthesis. The bridging ligand shared in the precursor successor and activated complex may or may.
Those in which electron transfer leaves each of the reaction partners in a stable oxidation state. Inner Sphere Electron Transfer. 2 Transfer can be a two step process from metal to ligand then ligand to metal.
The kinetics of the inner-sphere electron transfer reaction between a gold electrode and CO 2 was measured as a function of the applied potential in an aqueous environment. In an outer-sphere electrode reaction electron transfer between the electrode and the oxidantreductant takes place at the OHP while in an inner-sphere electrode reaction a coordinated ligand of electroactive metal complexes is bound to the electrode surface and. Outer sphere electron transfer occurs between complexes that do not undergo substitution.
Taube was awarded the Nobel Prize in chemistry in 1983. Inner Sphere Electron Transfer Mechanism - YouTube. La transferencia de electrones de la esfera interna IS ET o la transferencia de electrones enlazados es una reacción química redox que procede a través de un enlace covalente una fuerte interacción electrónica entre el oxidante y los reactivos reductores.
A brief intro to inner sphere electron transfer mechanismFollow me on Unacademy. Drawn between electron transfer in metal complexes and electron transfer in metalloenzymes. The award was based on his work on the mechanism of electron transfer reactions.
Inner sphere and outer sphere mechanisms are two different types of electron transfer mechanisms. The award was based on his work on. Inner sphere electron transfer IS ET or bonded electron transfer is a redox chemical reaction that proceeds via a covalent linkagea strong electronic interactionbetween the oxidant and the reductant reactants.
Inner sphere electron transfer and Antimony tetroxide See more Bridging ligand. Electron transfers that occur via ligands shared by the two metals undergoing oxidation and reduction are termed inner sphere electron transfers. Extraction of the electron transfer rate constant requires deconvolution of the current associated with CO 2 reduction from the competing hydrogen evolution reaction and mass transport.
The key difference between inner sphere and outer sphere mechanism is that the inner sphere mechanism occurs between complexes via a binding ligand whereas outer-sphere mechanism occurs between complexes that do not undergo substitution. Inner Sphere Electron Transfer Mechanism In this mechanism the electron is transferred from the reductant to oxidant through a bridge activated complex. A question this report aims to address is whether or not the MCR can predict rate constants for inner sphere electron transfer ISET reactions between iron complexes and O 2.
Taube was awarded the Nobel Prize in chemistry in 1983. Inner sphere electron transfer is very sensitive to bridging ligand. In addition to outer sphere reductionoxidation of O 2 O 2 the MCR is also applicable for proton transfer PT proton-coupled electron transfer PCET and hydride transfer rate constants.
While substitution reactions can be discussed without concern for oxidation-reduction reactions the reverse is not true.
Why Does The Inner Sphere Gets Some Charge Initially In A System Of Two Concentric Spheres Where The Outer Sphere Is Given A Charge Q While The Inner Sphere Is Earthed
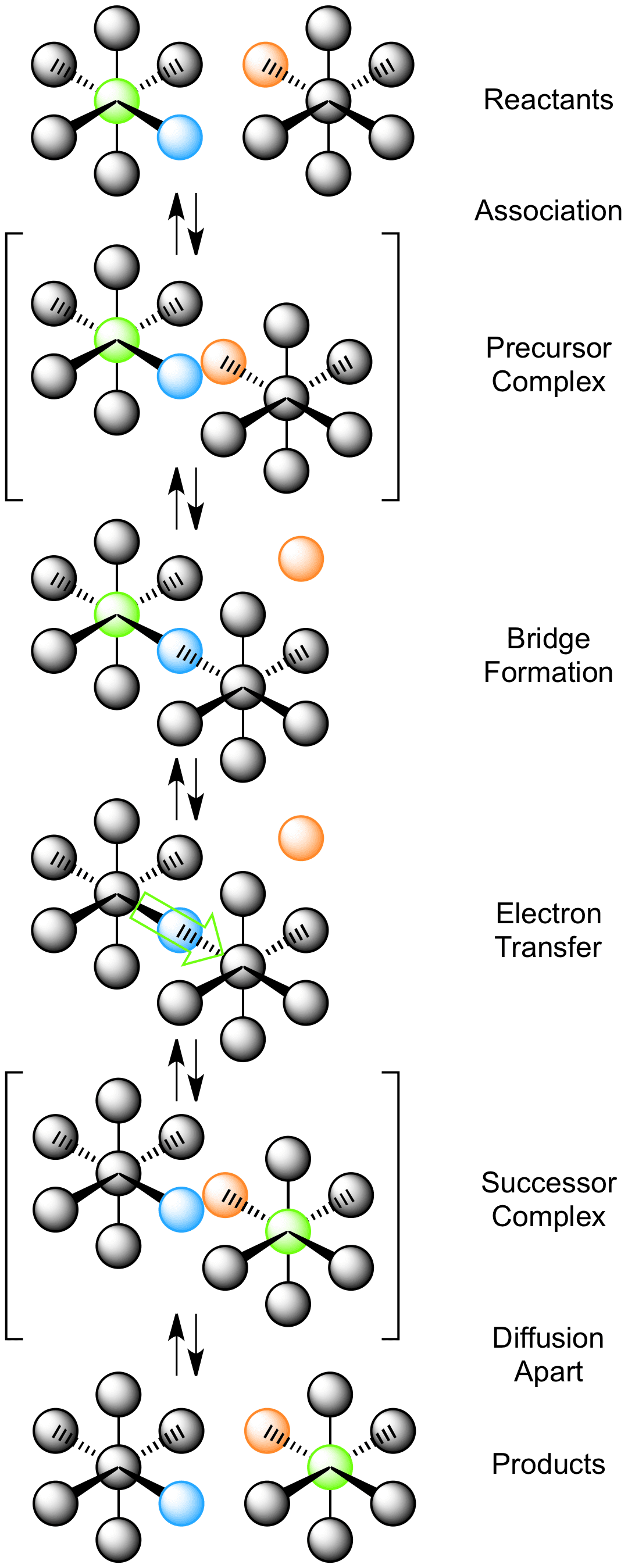
Inner Sphere Electron Transfer Mechanism

Suzuki Reaction And Stille Reaction Question From Csir Net And Gate Chemistry Organic Chemistry Reactions

30 Dank Science Memes For The Brainiacs Science Memes Memes Science

Molecular Orbital Theory And Bond Order Molecular Electron Configuration Theories
Learn About Electron Transfer Chegg Com

6 23 Inner Sphere Electron Transfer Youtube

3 A Inner Sphere Electron Transfer Process B Outer Sphere Electron Download Scientific Diagram

Mediated Inner Sphere Electron Transfer Induces Homogeneous Reduction Of Co2 Via Through Space Electronic Conjugation Hooe Angewandte Chemie International Edition Wiley Online Library
Inner Vs Outer Sphere Electron Transfer Mechanisms

Geometrical Isomerism In Coordination Compounds Organic Chemistry Study Geometric Chemistry

3 A Inner Sphere Electron Transfer Process B Outer Sphere Electron Download Scientific Diagram

Wagner Meerwein Rearrangement Important Points Examples Questions From C Wagner Daily Practices Pointe

Nomenclature Of Coordination Compounds Chemistry Compounds
